Loncar Lyon Jenkins has aggressively taken on “Big Pharma” since 1999. We’ve helped thousands of individuals across America who were injured by the negligence and greed of some of the largest drug manufacturers in the world.
Most pharmaceutical litigation involves the manufacturer’s failure to warn users of side-effects. Knowledge is power and if consumers know the true side-effects of certain medications manufacturers fear they would seek safer, equally effective alternatives.
On rare occasions, companies manipulate data to boost sales or protect patents. This is the case with the widely used medication Truvada.
Approved in 2001 this drug was originally sold under the brand name Viread. Over time, Viread was combined with other HIV medicines and sold under additional brand names, including Truvada, Symfi Lo, Cimduo, Genvoya, Atripla, Stribild and Complera – collectively referred to as “TAF” medications. Gilead Science’s TAF medications revolutionized how HIV was combatted, providing a once-a-day pill (pre-exposure prophylaxis, or PrEP). Truvada can be highly effective when used as a part of a regime to limit HIV exposure. However, this effectiveness carries a price beyond copays.
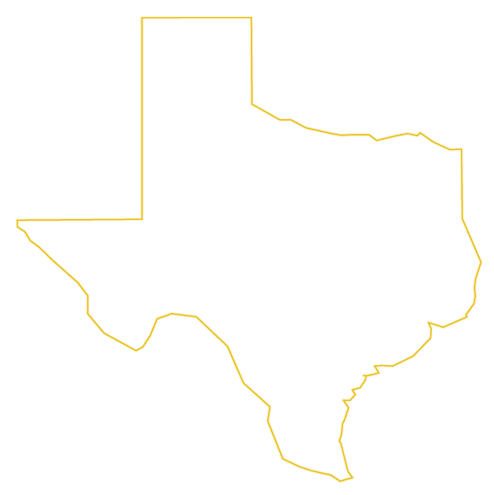
The links between bone breaks, kidney damage and TAFs in general and Truvada specifically are well-established and date back to the early 2000s.
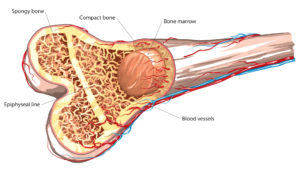
In this litigation, Gilead Science is accused of intentionally suppressing and manipulating clinical trial data to delay the release of information to the public concerning bone breaks and kidney damage. It is alleged that this data was intentionally kept from the public in order to protect sales of Truvada, while Gilead worked to develop a replacement, Descovy, which does not carry the same risks of bone and kidney injury. According to recent court filings, Gilead knew of the risk for bone loss and kidney damage in their medications as far back as 2000 but did not want to release safer alternatives until after the patent on Truvada expired.
Pharmaceutical manufacturers have a legal and moral duty to provide consumers with accurate information concerning the side-effects of their medication.

800-285-HURT (4878)Available 24/7 | 356 Days | se habla español
While many consumers would have still chosen to use Truvada despite the risks, that choice was never given to them. Instead, Gilead decided to make the choice for them. Perhaps, even more reprehensible, it is alleged that Gilead intentionally sought to delay the release of the safer alternative, Descovy, until after the patent on Truvada expired in 2021- simply to maintain market-share and profits.
Loncar Lyon Jenkins has successfully resolved cases for thousands of Americans unnecessarily injury by the greed of the pharmaceutical industry. We are glad that Truvada has been a revolutionary step-forward in combatting HIV/AIDS but that is no excuse for preventing safer, equally effective medications from reaching Americans.
Time is running out to file suit to be fairly compensated.
If you, or somone you know has used Viread, Truvada or any of the other versions of these PrEPs and were diagnosed with suspicious bone fractures or kidney disease while using these medicines, you need to act quickly. You may have a case that qualifies for significant compensation.
Contact Loncar Lyon Jenkins at 800-285-4878 or by completing the Contact Form for a confidential case evaluation. It costs you nothing unless we can collect a recovery on your behalf by jury verdict or settlement. Give us a call and let us fight for you.